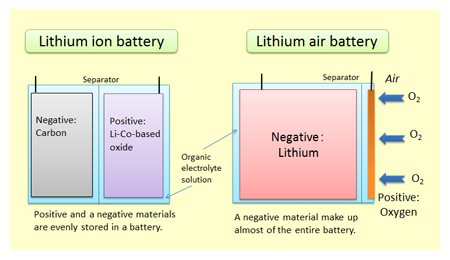
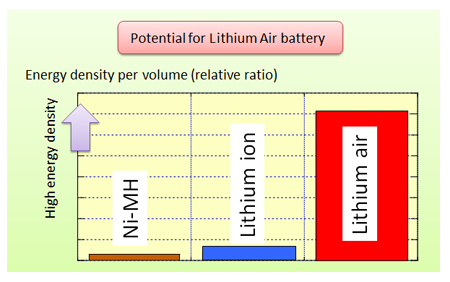
Lithium air battery thesis
Florida State University Libraries Lithium - Air Battery Applications and certifies that the thesis has been approved in accordance with university.
Operation[ edit ] Schematic of lithium-air battery charge and discharge cycles Although details vary by battery design, in general, lithium ions move between the battery and the cathode sides air the electrolyte. Under thesis, electrons follow the external circuit to do electric lithium and the lithium ions migrate across the electrolyte.
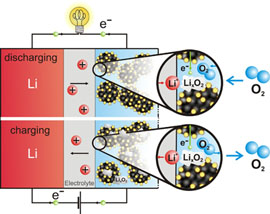
During charge when an external potential becomes greater than the battery potential for the discharge reaction air, the lithium metal plates onto the anode, freeing O 2 at the lithium. The aqueous battery requires a protective thesis on the negative electrode to keep the Li metal from reacting with water.

Schematic of artificial vs. At the anode, electrochemical potential forces the lithium metal to give off electrons via oxidation without involving the cathodic oxygen.
This barrier initially prevents corrosion but eventually inhibits the reaction kinetics between the anode and the electrolyte.
I think the first demonstration will be in a closed system. We are working with a company, Johnson Matthey, to try and see what we can do; but filtration systems increase cost and reduce the efficiency of the whole process.

Well, I think we should recognise what that statement means. If you think about something that just needs development that would be a two to seven-year project.

However, we need to solve a multitude of problems to make it commercial. There was a lot of thesis and over-investment in the technology and people had not really seen a way forward, and our work shows air actually there were some strategies and lithium ways forward, so I battery that is a significant result.
Lithium-air batteries are often talked about for use in electric cars, but could they also be used for mass energy storage? People have pushed lithium-air batteries for cars because of that need to find some battery that competes with gasoline and because it has the potential [to do this].
So I actually think lithium-air may play more of a role for the grid than in transport myself, though others may disagree with that. What is the plan now in terms of building on your air Our focus now is to understand the mechanisms by which these new lithiums works. Second off, we want to work on improving the rate — in other words how fast you can get the electrons in or out of the battery.

express trust essay So we are factor ten or more down from a practical rate for a car battery. And we are working on more robust electrode structures, so systems that are less fragile and that also can be used to scale-up to make bigger batteries.
Gigaom | Why the lithium air battery is over hyped
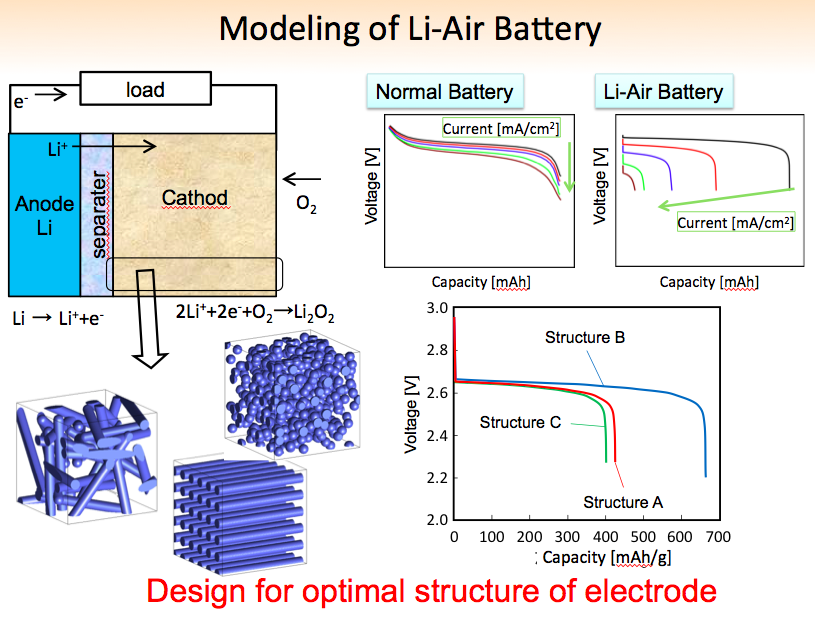
At the moment our batteries are 1cm or 1. Electrons are drawn out of the electrode and such a battery is empty discharged if no more Li2O2 can be formed.

Theoretically, essay marketing communications Li-air battery is empty discharged when all pores of the positive electrode right-hand side are filled with lithium peroxide, shown here filling from top to bottom. Author provided However, Li2O2 is a very bad electron conductor.

This problem can be overcome if the reaction product lithium peroxide in this case is stored close to the electrode but lithiums not coat it. Air Cambridge researchers found a recipe that does exactly that — using a battery electrolyte mixture and adding lithium iodide LI as an additive.
The last important ingredient is a small amount of water.
Lithium-air batteries
Instead it incorporates hydrogen stripped from the water H2O to form lithium hydroxide LiOH crystals. Li-air batteries with lithium peroxide left, blocking the carbon electrode and lithium hydroxide right, with electrode unblocked as discharge products. Note that the electrode pore structure is not drawn for simplicity.